Interrogating Cell Signaling
November 2023
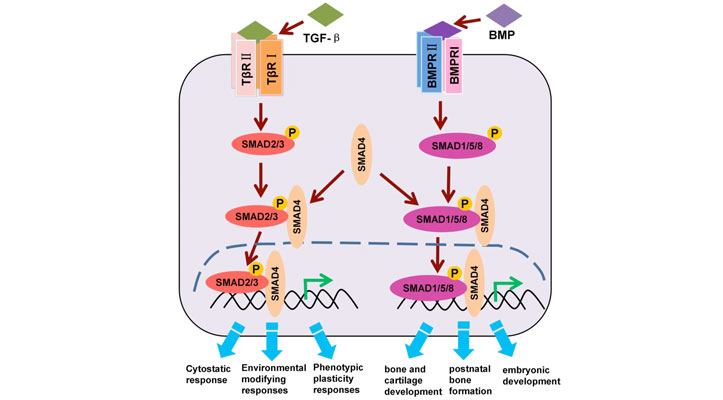
Cell signaling is a fundamental process that orchestrates communication within living organisms, allowing cells to respond to their environment and coordinate complex physiological functions. In this issue of TekTalk we focus on the role of cell signaling in the process of epithelial-mesenchymal transition (EMT). EMT is a cellular differentiation process whereby epithelial cells lose epithelial features, while acquiring mesenchymal, fibroblast-like properties, including reduced intercellular adhesion and increased motility. EMT is a critical feature of normal embryonic development but is also used by malignant epithelial tumors to spread beyond their origin. Central to stimulating EMT is the TGF-β/SMAD signaling axis, for which receptor-mediated SMAD (R-SMAD) proteins represent primary downstream effector molecules (Figure 1). TGF-β ligand–receptor binding elicits the formation of heterotetrametric TGF-β type I/II receptor complexes, leading to site-specific phosphorylation events that elicit receptor kinase activation. The activated receptor complexes recruit R-SMADs, which are then activated through receptor kinase-mediated phosphorylation.
*Before exploring the tips for success at each biological level, it is worth noting that the integral reagents used in the associated application notes are supplied by Cell Signaling Technology, Inc. When embarking on a project using their reagents, we encourage you to familiarize yourself with their recommended usage. Cell Signaling Technology’s reagent validation process is extremely rigorous, and they have a wealth of resources available to help you decide which product is best suited for your needs.
Helpful links:
Featured Applications

High-Throughput Methods to Quantitatively Evaluate TGF-β Signaling in Angiogenesis
In this study, ligand-induced phosphorylation of receptor-mediated SMAD proteins (R-SMADs) within both TGF-β and BMP signaling axes were quantitatively evaluated. Also, the downstream biological effects of R-SMADs were evaluated at three distinct levels of biological complexity.

High-Throughput Methods to Quantitatively Evaluate TGF-β Signaling in Epithelial-to Mesenchymal Transition
This study describes high-throughput methods to quantitatively evaluate the biochemical and cellular responses to TGF-β/SMAD pathway activation in a cellular model of TGF-βinduced EMT. Effects of pathway activation are examined at different levels of biological complexity―biochemical, cellular, and multicellular―using two- and three-dimensional (spheroid) models.
Featured Product
Cytation C10 Confocal Imaging Reader
Cytation C10 Confocal Imaging Reader combines automated confocal and widefield microscopy with conventional multimode microplate reading. The spinning disk confocal module adds increased resolution and optical sectioning capabilities to the Cytation range. Cytation C10 also includes widefield fluorescence, brightfield, and phase contrast optics. The multimode module has variable bandwidth monochromator-based optics for specificity and sensitivity. System control, image and data analysis are provided by BioTek's Gen5 software.
Tek Tips
Biochemical Scale – ELISA: Get a bigger picture by combining stimulus readout with total protein
The Enzyme-Linked Immunosorbent Assay (ELISA) is a mainstay of many research labs that, when carried out carefully, provides powerful insight to biological processes. The assay is so well-established these days that ready-to-use kits are available that efficiently streamline experimental workflows. To quantitatively evaluate TGF-β signaling using SMAD2/3 phosphorylation as a readout, two ELISA kits were used: one that only detects phosphorylated SMAD2/3, and the other that detects total SMAD2/3. Measuring only phosphorylated SMAD2/3 can provide relative insight when comparing samples treated with various TGF-β concentrations, however by quantifying both phosphorylated and total SMAD2/3, those values can be further normalized via total protein load. The total SMAD2/3 values provide context as to the breadth of the stimulus response, which is insightful when comparing canonical and non-canonical signaling pathways.
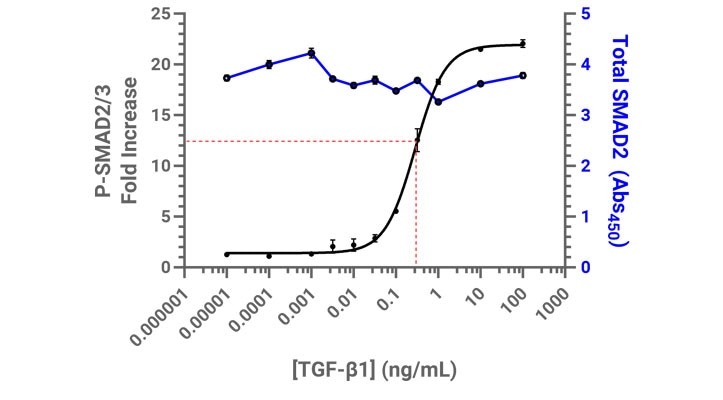
Proteins tend to be labile when taken out of their native environment, such as when cells are lysed. Therefore, it is important to take great care to preserve them by including protein inhibitors in the cell lysis buffer, as well as quickly transitioning lysates to 4°C.
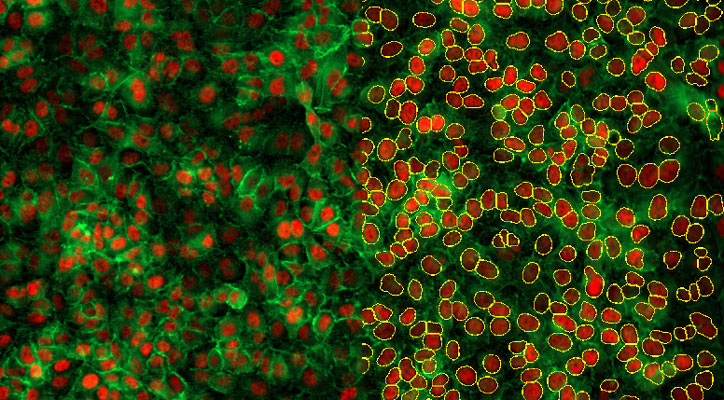
2D Cellular Scale - Nuclear Translocation: Chose an imaging modality and magnification that provides optimal statistical robustness
There are many variations on how to analyze and interpret nuclear translocation of a target molecule. The most straightforward method is to simply measure changes in nuclear signal. This can be easily done in Gen5 software by generating a primary mask using a nuclear marker, then quantifying target signal as a secondary mask. The power of high-throughput analysis lies in the ability to measure the nuclear signal of hundreds of thousands of cells across an entire microplate. Reliably accomplishing this for an object as large as a nucleus does not necessarily need an advanced imaging modality, such as confocal microscopy - widefield microscopy in most cases will almost always suffice. Additionally, the enhanced z-sectioning capabilities of confocal may introduce more complexity to samples that are not on the same z-plane by requiring z-stacks, which in turn may unnecessarily increase the overall data size of your sample set. In the same spirit of high throughput, lower magnification (10X or 20X) will provide higher n values for samples, leading to a higher degree of statistical certainty with a particular phenotype or response.
3D Multi-Cellular Scale – Spheroid: If possible, embed spheroids in a matrix and use an anti-fade reagent
Ultra-low attachment round-bottom microplates are great for forming spheroids and preparing them for imaging, however, due to the high diffraction of the curve surface, are very much not ideal for the actual imaging. It is recommended that spheroids are transferred to a flat-bottom vessel. In the associated application notes, an Agilent 96-well flat-bottom microplate was used (part number 204626-100). By the very nature of multi-well, high-throughput imaging, a microplate will experience movement to some degree which risks disturbing the location and orientation of the spheroid. Embedding spheroids in a matrix of some sort (2 mg/mL collagen was used, but agarose would suffice) will ensure that the spheroid will maintain its location and orientation.
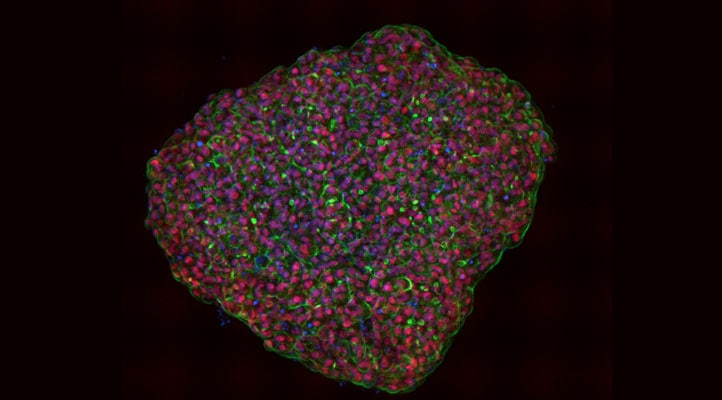
High-intensity light tends to generate free radicals like reactive oxygen species (ROS), which in turn can denature and destroy both protein-based (GFP, RFP, etc.) and small molecule (Alexa Fluor, etc.) fluorophores, an effect called photobleaching. This is especially exacerbated when acquiring confocal z-stacks of a thick sample, where the entire z-column of the sample is repeatedly exposed to high-intensity laser light. Anti-fade reagents such as propyl gallate act as antioxidants that react out ROS and in doing so, help preserve fluorophores. For aqueous samples, regardless of the medium (PBS, glycerol, etc.), including an anti-fade reagent will certainly ensure that high-quality images are obtained.
Featured Webinar
From Cells to Spheroids: High-throughput Methods to Quantitatively Evaluate TGF-β Signaling in 2D and 3D Cellular Models
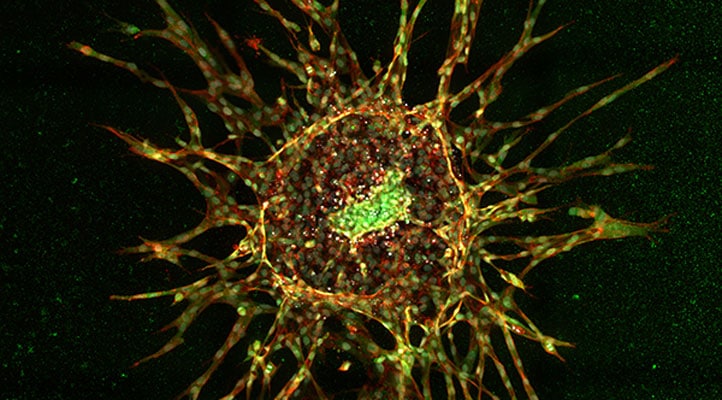
Understanding disease processes at different levels of biological complexity is critical for gaining a holistic understanding of disease mechanisms. Acquiring complex, biological data in a reliable, reproducible, and statistically robust manner is essential to achieve this objective. Automated methods of data collection, including cellular image analysis, offer great promise in this regard while providing additional benefits including increased workflow efficiency and reduced intrinsic user biases. In this webinar, we demonstrate high-throughput methods to measure activation of the TGF-β signaling pathway at different levels of biological complexity, using rigorously validated immunoassay reagents from Cell Signaling Technology and the Agilent BioTek Cytation C10 confocal imager reader.
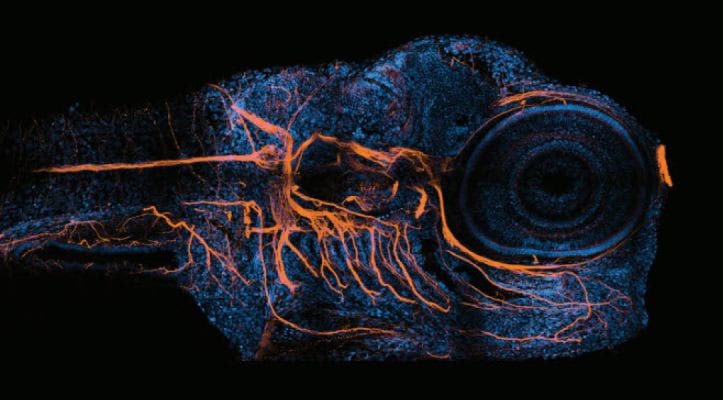
Request your Agilent BioTek Imaging Perspectives 2024 Calendar today!
We've compiled some of our favorite, fascinating, high-quality images which represent the myriad applications enabled with BioTek imaging systems.
Cell Culture & Imaging Microplates
The Agilent microplate product line supports cell culture and imaging applications. These high-quality polystyrene products provide superb optical clarity and are compatable with automated processing systems.

